DOCK Blaster:Tutorial 4: Difference between revisions
No edit summary |
|||
| (3 intermediate revisions by 3 users not shown) | |||
| Line 1: | Line 1: | ||
'''DOCK Blaster Tutorial 1. Docking Methotrexate (MTX) to Dhihydrofolate Reductase (DHFR)''' A[[DOCK Blaster:Tutorials | DOCK Blaster Tutorial]]. | |||
{{TOCright}} | |||
[[Image:1dfr.jpg|thumb|right|Mineralocorticoid receptor, PDB 1drf]] | |||
= Specific Aims = | |||
This tutorial will show you how to retrospectively dock a crystallographically observed ligand, aldosterone, into the human mineralocorticoid receptor, PDB code | |||
[http://www.rcsb.org/pdb/cgi/explore.cgi?pdbId=2AA2 2aa2]. You will also dock a list of annotated actives from the literature, as well as property-matched decoys. Finally, you will prospectively dock the ZINC "fragment-like" library into MR, and pick compounds to test as possible binders. The questions are: | |||
* 1. Can DOCK Blaster re-dock the native ligand close to its crystallographically observed position, with a competitive score? | |||
* 2. Can DOCK Blaster enrich known actives from a database of property-matched decoys? | |||
* 3. Can DOCK Blaster suggest novel, commercially available ligands for MR? | |||
[[Image:Mtx.png|thumb|right|Methotrexate, the crystallographically observed ligand of DHFR in 3dfr]] | |||
= Background and Significance = | |||
Methotrexate (MTX) binds to dihydrofolate reductase (DHFR) with high affinity, and has thus been used in chemotherapy for many years. DHFR reduces dihydrofolic acid to tetrahydrofolic acid, using NADPH as electron donor, which can be converted to the kinds of tetrahydrofolate cofactors used in 1-carbon transfer chemistry. Because tetrahydrofolate, the product of this reaction, is the active form of folate in humans, inhibition of DHFR can cause functional folate deficiency. Because folate is needed by rapidly dividing cells to make thymine, this effect may be therapeutic. | |||
= Preliminary Results = | |||
We will attempt to use DOCK Blaster to answer these questions, using data that has been prepared in advance. If you wish to use DOCK Blaster on your own project, you must prepare the data yourself. See [[DOCK Blaster:Input Preparation | Input Preparation]] for details. | |||
== Protocol 1 - Prepare input files == | |||
* P1.1. Go to the benchmarking data page [http://data.docking.org/ data.docking.org] and browse the DUD40 set, selecting the [http://data.docking.org/dud40/mr/ MR set]. Download each of these five files. On windows, you do this by right mouse clicking and selecting "save link as". If you are using your own target, you must prepare the files yourself (see [[DOCK Blaster:Input Preparation | Input Preparation]]). | |||
[[Image:DBtut1_1f.jpg|thumb|right|Figure T1.1 - Prepare input and launch job]] | |||
== Protocol 2 - Submit job and review results == | |||
* P2.1. Go to the [http://blaster.docking.org | DOCK Blaster main page] and select [http://blaster.docking.org/start.shtml | Queue A Job] from the DOCK! pull down menu (Figure T1.1). Fill in the form as in the figure, as follows. | |||
** a. In the "Target" field, select the receptor, rec.pdb. | |||
** b. In the next field, select "Docked ligand" and select the ligand, xtal-lig.mol2. | |||
** c. In the "Actives" field, select the actives.smi file. | |||
** d. In the "Inactives" field, select the inactives.smi file. | |||
** e. In "Email for reports", enter your email address (optional). | |||
** f. In the "Aim of this experiment" field, write a short memo about what you are doing. | |||
** g. Check your input, and click "DOCK!" when you are ready. | |||
If you have followed the steps above, your screen should now look something like Figure Tut1-1 (right). | |||
[[Image:DBtut1_2f.jpg|thumb|right|Figure T1.2 - Scrutinizer - data are uploaded, checked, and the job is started]]. | |||
* P2.2. Depending on available system resources, your job is now either running, or queued to run when a slot becomes available. If your screen does not look like Figure T1.2, check the input and try again. If the job is still failing after the third attempt, please write to support at docking.org for assistance. | |||
* P2.3. Read what is reported by the Scrutinizer (Figure T1.2), and proceed to the Job Watcher by clicking on the link at the bottom of the page when you are ready. | |||
* P2.4. You should now be in the Job Watcher (Figure T1.3). Depending on how much time has elapsed, and how busy our cluster is, you will see a progress report of the current status of your job. As time passes, the yellow status indicator should progress from left to right indicating the progress of your job through Target Preparation and Calibration. | |||
* P2.5. If all goes as usual, your job will have completed within an hour. You may reload the Job Watcher at any time to get an updated status report of your job. When calibration is complete, you may review the preliminary results and decide on the next course of action. | |||
* P2.6 Review Calibration results. blah blah blah. | |||
[[Image:DBtut1_3f.jpg|thumb|right|Figure T1.3]] | |||
= Proposed Research = | |||
Preliminary results above show that docking is able to enrich known actives from among a database of decoys. This suggests that prospective docking for discovery of novel ligands is reasonable and can be expected to suggest interesting compounds to test. | |||
== Protocol 3. Dock purchasable fragments == | |||
== Protocol 4. Review and interpret docking == | |||
== Protocol 5. Prioritize for testing == | |||
== Protocol 6. Suggested variations == | |||
== Possible problems & other approaches == | |||
[[Image:DBtut1_4f.jpg|thumb|right|Figure T1.4]] | |||
= Literature Cited = | |||
We refer you to our publications pages for theory and practice of docking and virtual screening. | |||
Document status: working tutorial. Please correct any errors you may find. | |||
[[Category:DOCK Blaster]] | |||
[[Category:Tutorials]] | |||
{{TOCright}} | |||
'''DOCK Blaster Tutorial 1. Docking Methotrexate (MTX) to Dhihydrofolate Reductase (DHFR)''' | |||
This is one of the [[DOCK Blaster:Tutorials]]. | This is one of the [[DOCK Blaster:Tutorials]]. | ||
== [[DOCK Blaster: | == Conceptual Overview == | ||
DOCK to a | Methotrexate (MTX, depicted) binds to human dihydrofolate reductase (DHFR) with high affinity, and has been used in clinical oncology for over 20 years. A crystal structure is available (PDB code 3dfr, depicted) with MTX in the binding site. | ||
* Question 1. Is DOCK Blaster able to suggest other compounds from a commercially available library that might be active against dihydrofolate reductase? | |||
** Sub-question 1.1. Is DOCK Blaster able to re-dock MTX close to its crystallographically observed position, with a competitive score? | |||
** Sub-question 1.2. Is DOCK Blaster able to enrich MTX compared with 50 property-matched decoys, compounds with similar properties to MTX but dissimilar topology? | |||
** Sub-question 1.3. Is DOCK Blaster able to enrich known actives, and de-enrich known inactives, against DHFR? | |||
picture of MTX | |||
picture of 3dfr | |||
picture of MTX bound to 3dfr | |||
picture of other DHFR ligands | |||
== From Theory to Action == | |||
We will attempt to use DOCK Blaster to answer these questions, using data that has been prepared in advance. If you wish to use DOCK Blaster on your own project, you must prepare data to conform with [[DOCK Blaster:Input Preparation | DOCK Blaster's]] basic requirements. To do this tutorial, please carefully follow the steps below. This tutorial usually takes about two hours, but most of the time is simply waiting for the computers to do the work on our servers. | |||
== Literature References == | |||
McMaster study | |||
Blaney's Magnum opus. | |||
== Collecting and formatting relevant data == | |||
== Launch Preliminary Calculation == | |||
** 1. Go to DOCK Blaster web page [http://blaster.docking.org blaster.docking.org]. Please note that during pre-alpha testing this page is different. Contact [[John Irwin]] for details. | |||
** 2. Click on "Start docking!" to go to the input preparation page. | |||
** 3. Click on "HELP" to open up the documentation for input preparation in a separate window. | |||
*** 3.1. In the documentation window, click on "Sample Data" at the end of the first paragraph. | |||
*** 3.2 In the sample data window, click on the link to data.docking.org | |||
*** 3.3. Click on DUD40. | |||
*** 3.4. Click on CDK2. | |||
*** 3.5. Right mouse to save all five files in this directory on your disk. | |||
*** 3.6. Close the documentation window, returning to the DOCK Blaster input form. | |||
** 4. Click on the first "Browse" button (to the right of Target) and select the rec.pdb file you just downloaded. | |||
** 5. In the next field "docked ligand" select the xtal-lig.mol2 file. | |||
** 6. Optionally, you may select the actives.smi and inactives.smi from the next two lines. | |||
** 7. Enter your email address if you wish to receive progress reports by email. | |||
** 8. Enter a brief comment about the calculation. | |||
** 9. (optional: enter cofactor.par below) | |||
If you have followed the steps above, your screen should now look something like Figure Tut1-1 (right). | |||
[[Image:Tut1-1.jpg|thumb|right|Figure Tut1-1]] | |||
** 10. Click on "DOCK" to upload the files and begin docking. You will be taken to the "Submission Scrutinizer", as depicted in Figure Tut1-2. | |||
[[Image:Tut1-2.jpg|thumb|right|Figure Tut1-2]] | |||
** 11. Your job should now be running. If it is not, there is either some part of the above instructions you did not follow, or we are currently experiencing problems with our systems. | |||
* | |||
** 12. Read through the submission scrutinizer, and go to the Job Watcher by clicking on the link at the bottom of the page... http://blaster.docking.org/cgi-bin/jobmon.pl | |||
** 13. You should now be in the Job Watcher. Depending on how much time has elapsed, and how busy our cluster is, you will see a progress report of your job. It might look something like Figure Tut1-3. [[Image:Tut1-3.jpg|thumb|right|Figure Tut1-3]] | |||
** 14. If all goes well, the job will complete in about an hour. You may reload the page at anytime to get updated status of the job progress. | |||
** 15. Preliminary docking normally begins after the site preparation is complete, often about 15 minutes after you first submitted the job. Once preliminary docking results are available, new links appear in the job watcher to allow you to start to see docked ligands. Caution: until the preliminary docking is complete, it can be misleading to rely upon incomplete preliminary docking results to reach any conclusions about the docking results. | |||
=== | == Examining the Results == | ||
== | == Docking a Database == | ||
== | == Browsing and interpreting the results == | ||
== Follow up and variations == | |||
== Possible problems and alternative approaches == | |||
[[Category:DOCK Blaster]] | [[Category:DOCK Blaster]] | ||
[[Category:Tutorials]] | [[Category:Tutorials]] | ||
Latest revision as of 03:23, 12 March 2014
DOCK Blaster Tutorial 1. Docking Methotrexate (MTX) to Dhihydrofolate Reductase (DHFR) A DOCK Blaster Tutorial.
Specific Aims
This tutorial will show you how to retrospectively dock a crystallographically observed ligand, aldosterone, into the human mineralocorticoid receptor, PDB code 2aa2. You will also dock a list of annotated actives from the literature, as well as property-matched decoys. Finally, you will prospectively dock the ZINC "fragment-like" library into MR, and pick compounds to test as possible binders. The questions are:
- 1. Can DOCK Blaster re-dock the native ligand close to its crystallographically observed position, with a competitive score?
- 2. Can DOCK Blaster enrich known actives from a database of property-matched decoys?
- 3. Can DOCK Blaster suggest novel, commercially available ligands for MR?
Background and Significance
Methotrexate (MTX) binds to dihydrofolate reductase (DHFR) with high affinity, and has thus been used in chemotherapy for many years. DHFR reduces dihydrofolic acid to tetrahydrofolic acid, using NADPH as electron donor, which can be converted to the kinds of tetrahydrofolate cofactors used in 1-carbon transfer chemistry. Because tetrahydrofolate, the product of this reaction, is the active form of folate in humans, inhibition of DHFR can cause functional folate deficiency. Because folate is needed by rapidly dividing cells to make thymine, this effect may be therapeutic.
Preliminary Results
We will attempt to use DOCK Blaster to answer these questions, using data that has been prepared in advance. If you wish to use DOCK Blaster on your own project, you must prepare the data yourself. See Input Preparation for details.
Protocol 1 - Prepare input files
- P1.1. Go to the benchmarking data page data.docking.org and browse the DUD40 set, selecting the MR set. Download each of these five files. On windows, you do this by right mouse clicking and selecting "save link as". If you are using your own target, you must prepare the files yourself (see Input Preparation).
Protocol 2 - Submit job and review results
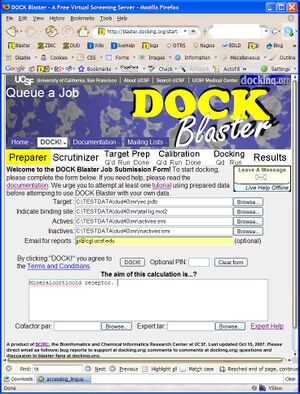
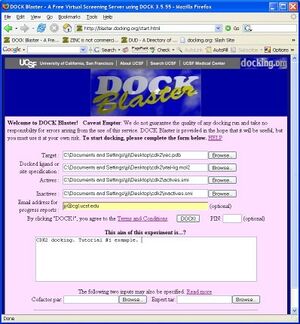
- P2.1. Go to the | DOCK Blaster main page and select | Queue A Job from the DOCK! pull down menu (Figure T1.1). Fill in the form as in the figure, as follows.
- a. In the "Target" field, select the receptor, rec.pdb.
- b. In the next field, select "Docked ligand" and select the ligand, xtal-lig.mol2.
- c. In the "Actives" field, select the actives.smi file.
- d. In the "Inactives" field, select the inactives.smi file.
- e. In "Email for reports", enter your email address (optional).
- f. In the "Aim of this experiment" field, write a short memo about what you are doing.
- g. Check your input, and click "DOCK!" when you are ready.
If you have followed the steps above, your screen should now look something like Figure Tut1-1 (right).
.
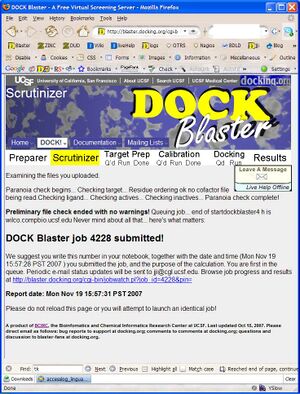
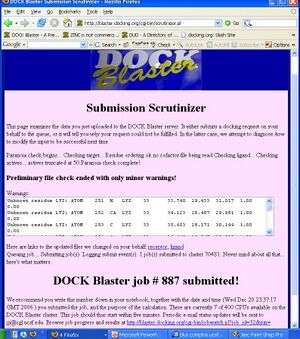
- P2.2. Depending on available system resources, your job is now either running, or queued to run when a slot becomes available. If your screen does not look like Figure T1.2, check the input and try again. If the job is still failing after the third attempt, please write to support at docking.org for assistance.
- P2.3. Read what is reported by the Scrutinizer (Figure T1.2), and proceed to the Job Watcher by clicking on the link at the bottom of the page when you are ready.
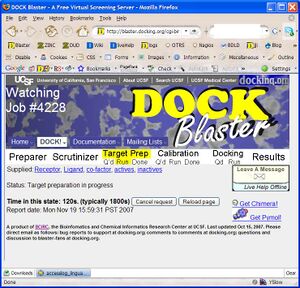
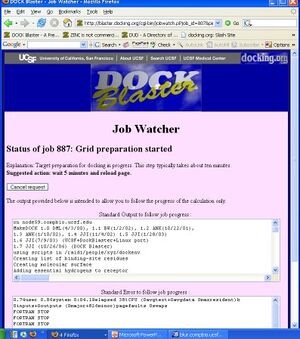
- P2.4. You should now be in the Job Watcher (Figure T1.3). Depending on how much time has elapsed, and how busy our cluster is, you will see a progress report of the current status of your job. As time passes, the yellow status indicator should progress from left to right indicating the progress of your job through Target Preparation and Calibration.
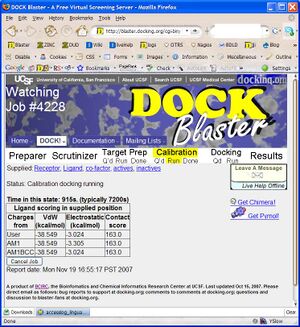
- P2.5. If all goes as usual, your job will have completed within an hour. You may reload the Job Watcher at any time to get an updated status report of your job. When calibration is complete, you may review the preliminary results and decide on the next course of action.
- P2.6 Review Calibration results. blah blah blah.
Proposed Research
Preliminary results above show that docking is able to enrich known actives from among a database of decoys. This suggests that prospective docking for discovery of novel ligands is reasonable and can be expected to suggest interesting compounds to test.
Protocol 3. Dock purchasable fragments
Protocol 4. Review and interpret docking
Protocol 5. Prioritize for testing
Protocol 6. Suggested variations
Possible problems & other approaches
Literature Cited
We refer you to our publications pages for theory and practice of docking and virtual screening.
Document status: working tutorial. Please correct any errors you may find.
DOCK Blaster Tutorial 1. Docking Methotrexate (MTX) to Dhihydrofolate Reductase (DHFR)
This is one of the DOCK Blaster:Tutorials.
Conceptual Overview
Methotrexate (MTX, depicted) binds to human dihydrofolate reductase (DHFR) with high affinity, and has been used in clinical oncology for over 20 years. A crystal structure is available (PDB code 3dfr, depicted) with MTX in the binding site.
- Question 1. Is DOCK Blaster able to suggest other compounds from a commercially available library that might be active against dihydrofolate reductase?
- Sub-question 1.1. Is DOCK Blaster able to re-dock MTX close to its crystallographically observed position, with a competitive score?
- Sub-question 1.2. Is DOCK Blaster able to enrich MTX compared with 50 property-matched decoys, compounds with similar properties to MTX but dissimilar topology?
- Sub-question 1.3. Is DOCK Blaster able to enrich known actives, and de-enrich known inactives, against DHFR?
picture of MTX
picture of 3dfr
picture of MTX bound to 3dfr
picture of other DHFR ligands
From Theory to Action
We will attempt to use DOCK Blaster to answer these questions, using data that has been prepared in advance. If you wish to use DOCK Blaster on your own project, you must prepare data to conform with DOCK Blaster's basic requirements. To do this tutorial, please carefully follow the steps below. This tutorial usually takes about two hours, but most of the time is simply waiting for the computers to do the work on our servers.
Literature References
McMaster study Blaney's Magnum opus.
Collecting and formatting relevant data
Launch Preliminary Calculation
- 1. Go to DOCK Blaster web page blaster.docking.org. Please note that during pre-alpha testing this page is different. Contact John Irwin for details.
- 2. Click on "Start docking!" to go to the input preparation page.
- 3. Click on "HELP" to open up the documentation for input preparation in a separate window.
- 3.1. In the documentation window, click on "Sample Data" at the end of the first paragraph.
- 3.2 In the sample data window, click on the link to data.docking.org
- 3.3. Click on DUD40.
- 3.4. Click on CDK2.
- 3.5. Right mouse to save all five files in this directory on your disk.
- 3.6. Close the documentation window, returning to the DOCK Blaster input form.
- 4. Click on the first "Browse" button (to the right of Target) and select the rec.pdb file you just downloaded.
- 5. In the next field "docked ligand" select the xtal-lig.mol2 file.
- 6. Optionally, you may select the actives.smi and inactives.smi from the next two lines.
- 7. Enter your email address if you wish to receive progress reports by email.
- 8. Enter a brief comment about the calculation.
- 9. (optional: enter cofactor.par below)
If you have followed the steps above, your screen should now look something like Figure Tut1-1 (right).
- 10. Click on "DOCK" to upload the files and begin docking. You will be taken to the "Submission Scrutinizer", as depicted in Figure Tut1-2.
- 11. Your job should now be running. If it is not, there is either some part of the above instructions you did not follow, or we are currently experiencing problems with our systems.
- 12. Read through the submission scrutinizer, and go to the Job Watcher by clicking on the link at the bottom of the page... http://blaster.docking.org/cgi-bin/jobmon.pl
- 13. You should now be in the Job Watcher. Depending on how much time has elapsed, and how busy our cluster is, you will see a progress report of your job. It might look something like Figure Tut1-3.
- 14. If all goes well, the job will complete in about an hour. You may reload the page at anytime to get updated status of the job progress.
- 15. Preliminary docking normally begins after the site preparation is complete, often about 15 minutes after you first submitted the job. Once preliminary docking results are available, new links appear in the job watcher to allow you to start to see docked ligands. Caution: until the preliminary docking is complete, it can be misleading to rely upon incomplete preliminary docking results to reach any conclusions about the docking results.